(구)글로벌 핫이슈
방사선생물학
![[Cancer Res.] The p53/p21 complex regulates cancer cell invasion and apoptosis by targeting Bcl-2 family proteins.](/enewspaper/upimages/admin_20170518164038_R.png) 2017년 05월호
2017년 05월호
[Cancer Res.] The p53/p21 complex regulates cancer cell invasion and apoptosis by targeting Bcl-2 family proteins.KIRAMS/ 엄홍덕*
- 출처
- Cancer Res.
- 등재일
- 2017 Apr 4
- 저널이슈번호
- pii: canres.2098.2016. doi: 10.1158/0008-5472.CAN-16-2098. [Epub ahead of print]
- 내용
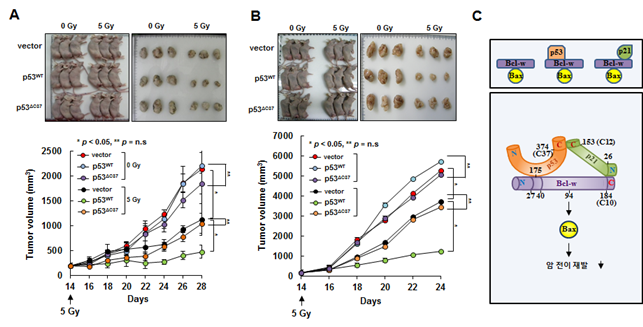
AbstractThe tumor suppressor p53 binds pro-survival Bcl-2 family proteins such as Bcl-w and Bcl-XL to liberate Bax, which in turn exerts pro-apoptotic or anti-invasive functions depending on stress context. Based on our previous finding that p53 interacts with p21, we investigated the possible involvement of p21 in these functions. Here we report that although p53 can bind Bcl-w alone, it requires p21 to liberate Bax to suppress cell invasion and promote cell death. p21 bound Bcl-w, forming a p53/p21/Bcl-w complex in a manner that maintained all pairwise p53/p21, p21/Bcl-w, and p53/Bcl-w interactions. This allowed Bax liberation from the complex. Accordingly, a p53 derivative incapable of binding p21 failed to mediate radiotherapy-induced tumor cell death in mice. Bcl-XL also served as a target of the cooperative action of p53 and p21. Overall, our findings indicate that the p53/p21 complex rather than p53 itself regulates cell invasion and death by targeting Bcl-2 proteins. We propose that the p53/p21 complex is a functional unit that acts on multiple cell components, providing a new foundation for understanding the tumor-suppressing functions of p53 and p21.
Author information
Kim EM1, Jung CH1, Kim J2, Hwang SG3, Park J4, Um HD5.
1Division of Applied Radiation Bioscience, Korea Institute of Radiological & Medical Sciences.2Laboratory of Radiation Tumor Physiology, Korea Institute of Radiological and Medical Sciences.3Division of Radiation Cancer Biology, Korea Institute of Radiological & Medical Sciences.4Lab of Radiation Cancer Biology, KIRAMS.5Division of Applied Radiation Bioscience, Korea Institute of Radiological & Medical Sciences hdum@kcch.re.kr.
관련자료 보기
- 연구소개
- p53 단백질은 암 전이를 억제하고 방사선을 비롯한 다양한 항암제들의 암세포 사멸 작용을 매개하는 가장 대표적인 암 억제인자로 알려져 있습니다. p53 단백질의 중요성은 전체 암환자의 절반가량이 암세포에 p53 유전자의 변이를 동반하며 이로 인하여 p53의 암 억제 기능이 제대로 발휘되지 못한다는 점으로도 인식할 수 있습니다. p53은 세포핵에서 전사인자로 작용하기도 하지만 세포질, 특히 미트콘드리아에서도 아주 중요한 암 억제 기능을 수행하는 것으로 알려져 있습니다. 이 후자의 경우, p53은 Bcl-2 계열 생존인자 (Bcl-2, Bcl-XL, Bcl-w 등) 들에 결합하며 그 결과 이 생존인자들에 부착되어 있던 Bcl-2 계열 사멸인자 (Bax, Bak)들을 자유롭게 풀어줍니다. 생존인자들로부터 풀려난 Bax와 Bak은 상황에 따라 암세포의 침윤과 전이를 억제하기도 하고 방사선이나 항암제의 암세포사멸 작용을 매개하기도 합니다. 본 연구진은 p53 단백질과 p21 단백질이 직접 결합함을 보고한 바 있습니다 (Kim J et al. EMBO Reports 15:1062-8, 2014). 이를 바탕으로 본 연구에서는 p21 단백질이 p53 단백질의 미트콘드리아에서의 암 억제 작용에 관여할 가능성을 Bcl-w와 Bax를 주 모델로 하여 다양한 암세포 (폐암, 대장암, 신경아세포종) 들을 분석하였습니다. 그 결과, p21은 p53의 미트콘드리아에서의 작용에 반드시 필요하며 그 기전은 p53, p21, Bcl-w 모두가 서로 상호간 접점을 형성하는 결합체를 형성하고 이러한 p53/p21/Bcl-w 결합체가 형성되어야만 Bcl-w에 부착되어 있던 Bax가 분리되어 암 침윤ㆍ전이가 억제되고 방사선치료 효과가 극대화됨을 확인하였습니다. 또한, 이러한 모델은 Bcl-w 에만 국한되는 것이 아니고 Bcl-XL과 Bcl-2에도 적용됨을 확인하였습니다. 본 연구를 통하여 p53/p21 결합체가 다양한 암 촉진인자들의 작용을 조절하는 하나의 기능체임이 확인되어 p53 연구와 암 전이ㆍ재발 연구에 새로운 전기를 마련했다고 평가됩니다.
- 덧글달기







