글로벌 연구동향
방사선종양학
![[Radiother Oncol] Clinical impact of combined transarterial chemoembolization and radiotherapy for advanced hepatocellular carcinoma with portal vein tumor thrombosis: An external validation study.](/enewspaper/upimages/admin_20160414163145_R.jpg) 2016년 04월호
2016년 04월호
[Radiother Oncol] Clinical impact of combined transarterial chemoembolization and radiotherapy for advanced hepatocellular carcinoma with portal vein tumor thrombosis: An external validation study.울산대학교,성균관의대 /윤상민*,박희철*
- 출처
- Radiother Oncol
- 등재일
- 2016 Feb
- 저널이슈번호
- 118(2):408-15. doi: 10.1016/j.radonc.2015.11.019. Epub 2016 Jan 28.
- 내용
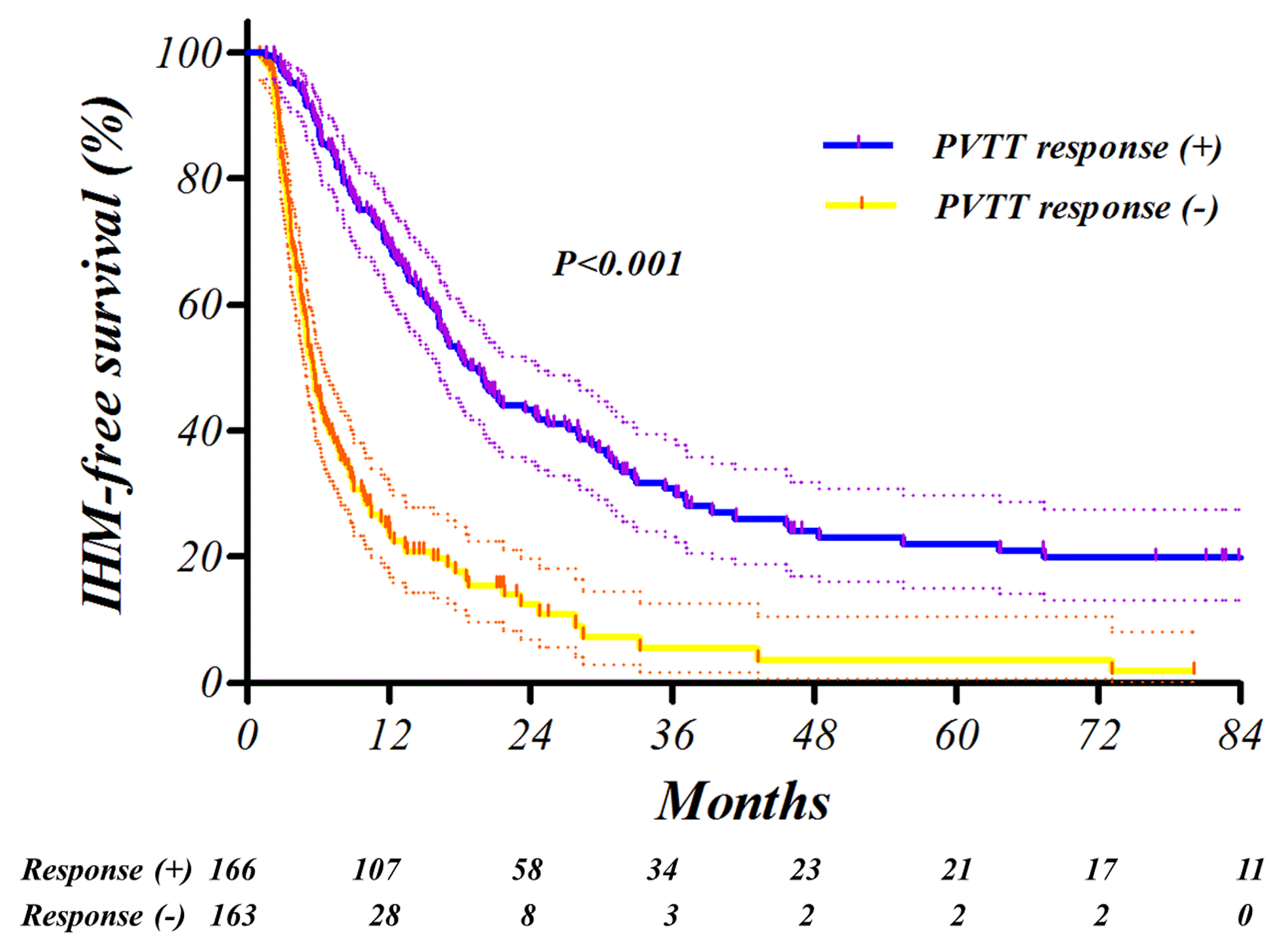
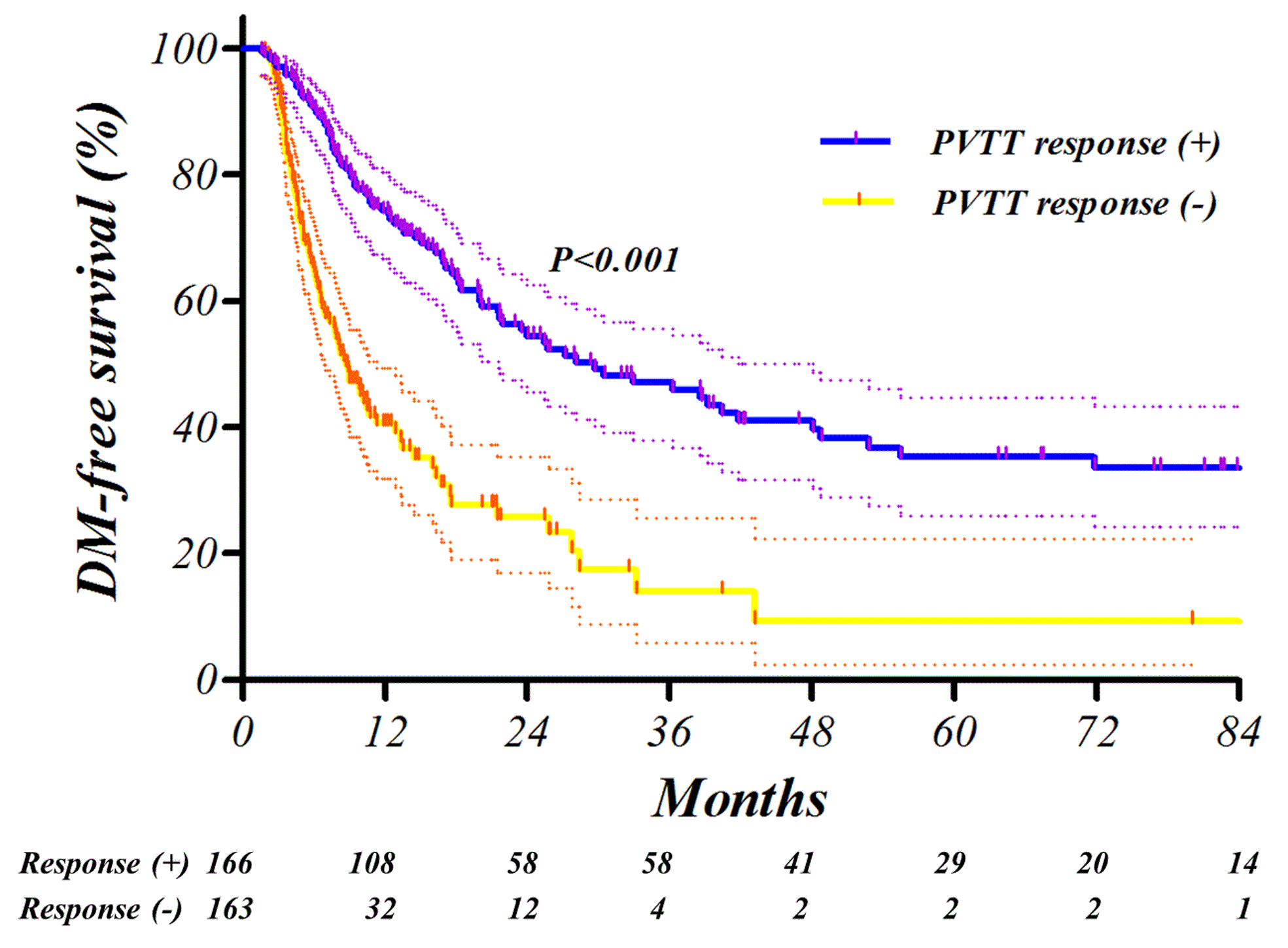
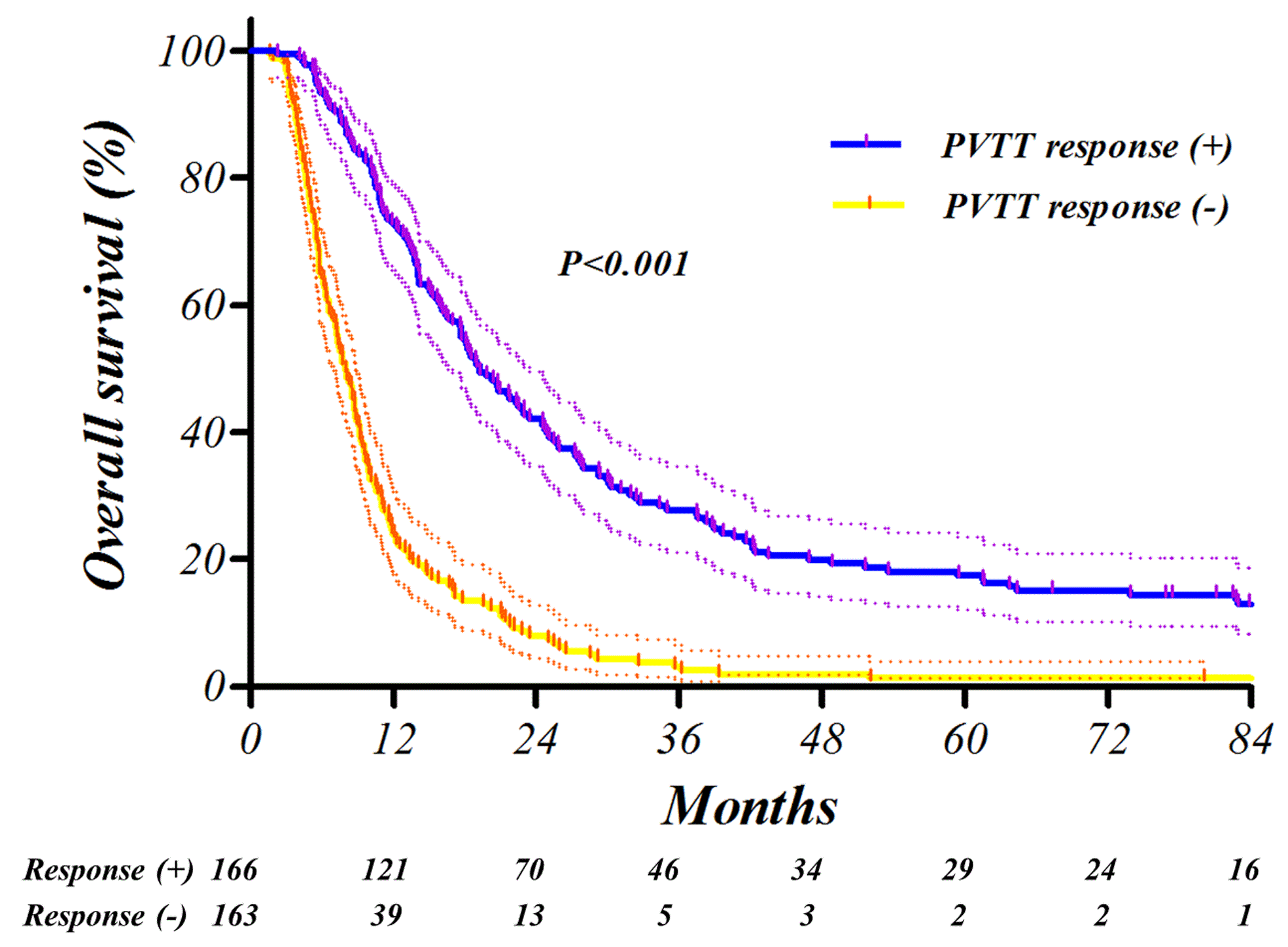
Fig. Kaplan-Meier survival curves. The intrahepatic metastasis-free survival (IHMFS, A), distant metastasis-free survival (DMFS, B) and overall survival (OS, C) were significantly affected by PVTT response in the training setAbstract
PURPOSE:
To evaluate the relationship between portal vein tumor thrombosis (PVTT) response and clinical outcomes in patients with hepatocellular carcinoma (HCC) treated with transarterial chemoembolization followed by radiotherapy (TACE-RT).
MATERIALS AND METHODS:
The study enrolled 329 patients in the training set and 179 patients in the validation set. All patients who were treated with TACE-RT from 2002 to 2008 and satisfied the inclusion criteria were enrolled in the study. The median follow-up period was 11.7months (range, 1.6-108.6) in the training set and 11.9months (range, 1.7-105.1) in the validation set.
RESULTS:
After TACE-RT, PVTT response was complete or partial in 32 (9.7%) and 134 (40.7%) patients of the training set and in 18 (10.1%) and 96 (53.6%) patients in the validation set, respectively. Failure to obtain PVTT response was significantly related with elevated post-treatment Child-Pugh score (P<0.001). Furthermore, progression-free survival was significantly related with PVTT response (P<0.001, hazard ratio 0.33, 95% confidence interval 0.25-0.42) in multivariate analysis. In receiver-operating characteristics analysis of 1-year progression prediction, the PVTT response showed an area under the curve of 0.74. Most of the findings were successfully reproduced in the independent external validation set.
CONCLUSIONS:
Positive PVTT response was closely associated with favorable clinical outcomes. The PVTT response to TACE-RT reduces metastasis and makes it possible to maintain normal liver function and achieve longer survival.
Author information
Yu JI1, Park JW2, Park HC3, Yoon SM4, Lim do H1, Lee JH5, Lee HC6, Kim SW7, Kim JH2.
1Department of Radiation Oncology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Republic of Korea.
2Department of Radiation Oncology, Asan Liver Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Republic of Korea.
3Department of Radiation Oncology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Republic of Korea; Department of Medical Device Management and Research, SAIHST, Sungkyunkwan University, Seoul, Republic of Korea. Electronic address: hee.ro.park@samsung.com.
4Department of Radiation Oncology, Asan Liver Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Republic of Korea. Electronic address: drsmyoon@amc.seoul.kr.
5Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Republic of Korea.
6Department of Gastroenterology, Asan Liver Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Republic of Korea.
7Biostatistics Team, Samsung Biomedical Research Institute, Seoul, Republic of Korea.
- 키워드
- Clinical impact of combined transarterial chemoembolization and radiotherapy for advanced hepatocellular carcinoma with portal vein tumor thrombosis: An external validation study.
- 연구소개
- 간문맥종양혈전증을 동반한 간세포암의 치료에서 표적치료제인 sorafenib이 표준치료법임에도 불구하고, 임상 현장에서는 간동맥화학색전술과 체외방사선치료를 병용하는 병용 요법이 많이 적용되고 있습니다. 그러나 이런 병용 요법의 활용에 근거를 제시하는 좋은 과학적 데이터는 제시되지 못하고 있던 현실입니다. 본 연구 논문은 간문맥종양혈전증에 체외방사선치료 후 치료 반응이 있으면, 치료 후의 간내 전이 및 간외 전이가 줄어들고, 이를 통해 생존율이 높아질 수 있다는 근거를 제시하고 두 대형병원의 데이터를 통해 확인한 의미가 있습니다.
- 덧글달기
- 이전글 [Radiother Oncol] Comparison of weekly versus triweekly cisplatin delivered concurrently with radiation therapy in patients with locally advanced nasopharyngeal cancer: A multicenter randomized phase II trial (KCSG-HN10-02).
- 다음글 [Radiother Oncol] A phase II study of preoperative mFOLFOX6 with short-course radiotherapy in patients with locally advanced rectal cancer and liver-only metastasis.







