글로벌 연구동향
방사선종양학
![[Int J Radiat Oncol Biol Phys] Concurrent Chemoradiation Therapy Followed by Consolidation Chemotherapy for Localized Extranodal Natural Killer/T-Cell Lymphoma, Nasal Type.](/enewspaper/upimages/admin_20151214162347_R.jpg) 2015년 12월호
2015년 12월호
[Int J Radiat Oncol Biol Phys] Concurrent Chemoradiation Therapy Followed by Consolidation Chemotherapy for Localized Extranodal Natural Killer/T-Cell Lymphoma, Nasal Type.성균관의대 / 안용찬*
- 출처
- Int J Radiat Oncol Biol Phys
- 등재일
- 2015 Nov 1
- 저널이슈번호
- 93(3):677-83. doi: 10.1016/j.ijrobp.2015.07.2267. Epub 2015 Jul 22.
- 내용
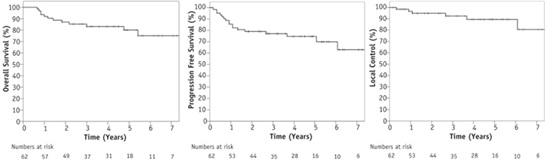
3년/5년 생존률 = 83.1%/80.1%; 3년/5년 무병생존률 = 77.1%/69.9%; 3년/5년 국소억제율 = 92.4%/89.5%.Abstract
PURPOSE:
To evaluate the effectiveness of concurrent chemoradiation therapy (CCRT) with 40 Gy followed by consolidation chemotherapy for localized extranodal natural killer (NK)/T-cell lymphoma (ENKTL), nasal type.METHODS AND MATERIALS:
From August 2004 to August 2012, 62 patients with newly diagnosed stage IE to IIE ENKTL underwent CCRT followed by consolidation chemotherapy. The median RT dose was 40 Gy. Cisplatin, 30 mg/m(2), was administered weekly during the RT course. Responders to CCRT were encouraged to undergo consolidation chemotherapy. Three different consolidation chemotherapy regimens were used consecutively: VIPD (etoposide, ifosfamide, cisplatin, and dexamethasone); VIDL (etoposide, ifosfamide, and dexamethasone followed by intramuscular injection of l-asparaginase); and MIDLE (methotrexate, etoposide, ifosfamide, mesna, and l-asparaginase).RESULTS:
The median follow-up period was 49 months (range 8-112). After completion of CCRT, 56 patients (90.3%) had a complete response, 4 (6.4%) had a partial response, 1 (1.6%) had stable disease, and 1 patient (1.6%) had progressive disease (PD). Consolidation chemotherapy was recommended to 61 patients, after excluding the patient with PD, but was actually delivered to 58. Of these 58 patients, 56 (96.5%) had a complete response and 2 (3.5%) had PD. During the follow-up period, 17 patients (including 3 with PD) experienced progression. The median interval to progression was 11 months (range 1-61). Local failure developed in 6 patients, of whom, 2 had developed progression outside the RT field. For all patients, the 3-year overall survival, progression-free survival, and local control rates were 83.1%, 77.1%, and 92.4%, respectively. Grade ≥3 nonhematologic toxicity developed in only 3 patients (4.8%).CONCLUSIONS:
Excellent clinical outcomes were achieved using CCRT with 40 Gy followed by consolidation chemotherapy. Additional investigation, however, is warranted to confirm our findings.Author information
Oh D1, Ahn YC2, Kim SJ3, Kim WS3, Ko YH4.
1Department of Radiation Oncology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
2Department of Radiation Oncology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. Electronic address: ycahn.ahn@samsung.com.
3Division of Hematology and Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
4Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 연구소개
- 비강(코) 및 주변에 많이 발병하는 NK/T 세포 림프종에 대한 최적의 치료법에 대해서는 아직 의사들 사이에 이견이 많이 있어 왔다. 연구자는 동시병용 항암화학요법 (concurrent chemoradiation therapy)를 근간으로 하는 치료방침을 적용하여 탁월한 임상결과를 얻어 보고한 바 있다. 우리가 채택한 방사선량은 44~45 Gy 수준이나 외국의 연구자들은 여전히 “50 Gy 이상의 방사선량을 주어야 한다”는 주장이 많았다. 본 연구는 동시병용 방사선-항암화학요법을 적용하면서 40 Gy의 방사선량을 채택하였고 탁월한 치료효과를 얻을 수 있다는 임상적 근거를 처음으로 제시하였다.
- 덧글달기
- 이전글 [Radiat Oncol] Definitive radiotherapy alone over 60 Gy for patients unfit for combined treatment to stage II-III non-small cell lung cancer: retrospective analysis
- 다음글 [J Neurooncol] Nomogram prediction of survival in patients with brain metastases from hepatocellular carcinoma treated with whole-brain radiotherapy: a multicenter retrospective study.







