글로벌 연구동향
방사선종양학
- 2021년 05월호
[J Hepatol.] Proton beam radiotherapy vs. radiofrequency ablation for recurrent hepatocellular carcinoma: A randomized phase III trial 재발성 간암에서 양성자치료와 고주파소작술의 무작위비교 임상시험국립암센터 / 김태현, 고영환, 박중원*
- 출처
- J Hepatol.
- 등재일
- 2021 Mar
- 저널이슈번호
- 74(3):603-612. doi: 10.1016/j.jhep.2020.09.026. Epub 2020 Oct 5.
- 내용
-
Abstract
Background & aims: Proton beam radiotherapy (PBT) has recently been applied to treat hepatocellular carcinoma (HCC); however, there is no randomized controlled trial-based evidence on its safety and efficacy. We compared the outcomes of PBT and radiofrequency ablation (RFA) in patients with recurrent/residual HCC (rHCC) in a phase III non-inferiority trial.Methods: Patients with rHCC (size <3 cm, number ≤2) were randomly assigned to receive PBT or RFA according to Child-Pugh score and tumor stage. After randomization, if the assigned treatment was technically infeasible, crossover was allowed. The primary endpoint was 2-year local progression-free survival (LPFS), with a non-inferiority margin of 15% in the per-protocol (PP) population; a complementary analysis was performed in the intention-to-treat (ITT) population (NCT01963429).
Results: The ITT population comprised 144 patients receiving either PBT (n = 72) or RFA (n = 72). Six patients switched from the PBT arm to the RFA arm and 19 patients switched from the RFA arm to the PBT arm. In the PP population, the 2-year LPFS rate with PBT (n = 80) vs. RFA (n = 56) was 94.8% vs. 83.9%, a difference of 10.9 percentage points (90% CI 1.8-20.0; p <0.001); in the ITT population, the 2-year LPFS rate with PBT vs. RFA was 92.8% vs. 83.2%, a difference of 9.6 percentage points (90% CI 0.7-18.4; p <0.001), meeting the criteria for non-inferiority. The 3- and 4-year LPFS rates for PBT were also non-inferior to those for RFA. The most common adverse events were radiation pneumonitis (32.5%) and decreased leukocyte counts (23.8%) for PBT and increased alanine aminotransferase levels (96.4%) and abdominal pain (30.4%) for RFA. No Grade 4 adverse events or mortality were noted.
Conclusions: PBT showed LPFS values that were non-inferior to those for RFA; in addition, PBT was tolerable and safe. CLINICAL TRIAL NUMBER: #NCT01963429 (ClinicalTrials.gov).
Lay summary: Radiofrequency ablation is the standard of care for patients with small hepatocellular carcinoma in whom surgery is not feasible. This study is the first phase III randomized controlled trial to evaluate the clinical outcomes of proton beam radiotherapy vs. radiofrequency ablation in patients with recurrent small HCC. Our findings show that this new technique is not inferior and can be applied safely in patients with small recurrent hepatocellular carcinoma.
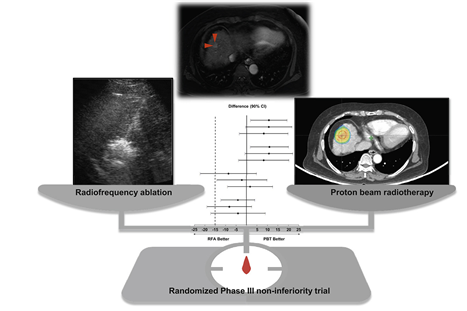
본 연구의 1차 목표인 2년 국소무진행생존 (local progression-free survival) Forest Plot으로서 교차치료 연구에서는 per protocol 분석이 우선이며 보조적으로 intention-to-treatment 분석도 시행하였는데 두 방법 모두에서 양성자치료가 더 우월하였다. 3년, 4년 분석에서도 비열등성 경계인 –15% 보다 충분히 적은 차이를 보여서 두 치교간의 차이가 없음을 알 수 있다.
Affiliations
Tae Hyun Kim 1 , Young Hwan Koh 2 , Bo Hyun Kim 3 , Min Ju Kim 4 , Ju Hee Lee 2 , Boram Park 5 , Joong-Won Park 6
1 Center for Liver and Pancreatobiliary Cancer, National Cancer Center, Goyang, Republic of Korea; Center for Proton Therapy, National Cancer Center, Goyang, Republic of Korea.
2 Center for Liver and Pancreatobiliary Cancer, National Cancer Center, Goyang, Republic of Korea; Department of Radiology, National Cancer Center, Goyang, Republic of Korea.
3 Center for Liver and Pancreatobiliary Cancer, National Cancer Center, Goyang, Republic of Korea.
4 Department of Radiology, National Cancer Center, Goyang, Republic of Korea.
5 Biostatistics Collaboration Team, Research Core Center, National Cancer Center, Goyang, Republic of Korea.
6 Center for Liver and Pancreatobiliary Cancer, National Cancer Center, Goyang, Republic of Korea. Electronic address: jwpark@ncc.re.kr.
- 키워드
- Hepatocellular carcinoma; Radiofrequency ablation: proton beam therapy; Randomised controlled trial; local progression-free survival.
- 연구소개
- 국립암센터 박중원 (소화기내과 간장학), 김태현 (방사선종양학과), 고영환 (영상의학과) 등의 연구팀은 2013년부터 7년간 전향적 무작위 대조군 3상 임상연구를 수행하여 간세포암종 환자에서의 양성자치료 효과를 밝히고, 연구성과를 간분야의 최고 학술지인 유럽간학회지(Journal of Hepatology, IF 20.582) 최신호 표지 논문으로 게재했다. 현재 간세포암종 환자에서 완치목적의 표준치료인 고주파열치료와 비교해 양성자치료가 안전하고 효과적임을 입증했다. 간세포암종에 양성자치료를 우리나라보다 먼저 적용하였던 선진국 서양이나 일본에서도 입증하지 못했던 치료효과를 비열등성 3상 임상시험을 통해서 세계 최초로 입증한 것이다. 3cm 이하의 초기 간세포암종은 일차적으로 수술적 치료가 권장된다. 그러나 수술이 여의치 않을 경우 시행되는 완치목적의 대표적 비수술적 표준치료가 고주파열치료이다. 고주파열치료는 간암을 고주파로 태워서 치료하는 국소치료법이다. 양성자치료는 이미 많은 간세포암종 환자들에게서 효과적임을 보여준 치료법이지만, 현재까지 표준치료와 비교하는 무작위 대조군 3상 임상연구가 없었다. 간세포암종 환자 144명 중 간기능등급과 병기에 따라 양성자치료군과 고주파열치료군으로 무작위 배정했다. 배정 이후 배정된 치료가 기술적으로 불가능한 경우 상호교차 치료를 허용했다. 실제 치료는 80명이 양성자치료를, 56명이 고주파열치료를 받았다. 그 결과, 2년 국소무진행생존율은 양성자치료군 94.8%, 고주파열치료군은 83.9%로 나타났다. 추가 관찰한 3년 및 4년 국소무진행생존율도 의미있는 차이가 없었다. 이로써 본 연구의 일차목표를 달성하였다. 또한, 고주파열치료가 불가능해 양성자치료를 받은 환자군이 반대의 경우보다 많았다. 고주파열치료군으로 배정된 환자 72명 중 22명(30%)은 고주파열치료가 불가능했고, 이 중 19명은 양성자치료가 가능했다. 이에 반해 양성자치료군으로 배정된 환자 72명 중 11명(15%)이 양성자치료가 불가능했고, 이 중 6명이 고주파열치료가 가능했다. 부작용 측면에서 두 치료법 간에 큰 차이가 없었다. 가장 흔한 부작용은 양성자치료군에서는 중증도 이하 방사선 폐렴(32.5%), 백혈구수감소(23.8%)가, 고주파열치료군에서는 알라닌아미노트랜스퍼라제 수치 증가(96.4%) 복통(30.4%)로서, 양성자치료는 심각한 부작용이나 사망 없이 안전한 치료임을 확인했다. 이 연구를 통해 양성자치료는 작은 간세포암종의 완치를 목적으로 하는 표준치료의 하나가 될 수 있음을 학술적으로 입증하였다.
- 덧글달기








편집위원
3cm 보다 작은 간세포암종에서 표준치료인 RFA와 비교하여 양성자치료를 활용한 hypofractionated RT의 비열등성을 보여준 최초의 3상 임상시험입니다.
덧글달기닫기2021-05-06 15:00:38
등록
편집위원2
재발 간암 환자에 대한 양성자와 RFA 치료 간의 임상적 유효성 비교에 대한 논문으로 임상적으로 환자 치료의 선택에 있어서 매우 유의미한 정보를 제공
덧글달기닫기2021-05-06 15:44:00
등록