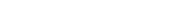글로벌 연구동향
방사선생물학
- [Int J Radiat Biol.] Targeting Chemo-Proton Therapy on C6 cell line using Superparamagnetic Iron Oxide Nanoparticles conjugated with Folate and Paclitaxel.
건양대/ 강성희,홍성표, 강보선 *
- 출처
- Int J Radiat Biol.
- 등재일
- 2018 Jul 23:1-30. doi: 10.1080/09553002.2018.14958
- 저널이슈번호
- 내용
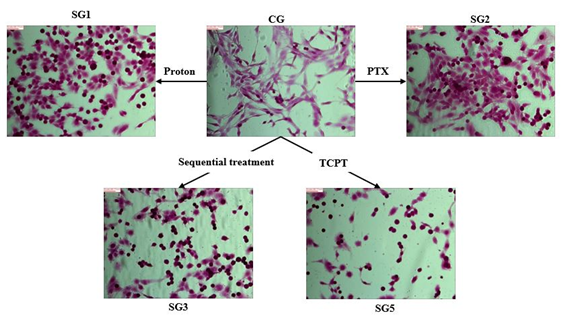
Figure 7. The morphologies of C6 cells. Upper row: control group (center), proton exposed group (left), PTX treated group (right). Lower row: proton exposure with sequential treatement of PTX group (left), TCPT treatment group (right). Proton exposure for the treatment was 8 Gy. Scale bar = 86.77 μm.
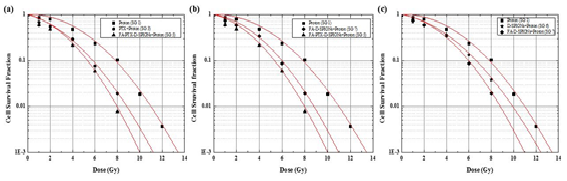
Figure 8. The results of colonogenic assays of C6 cells with different radiation doses and different treatment with PTX, D-SPIONs, FA-D-SPIONs, or FA-PTX-DSPIONs. The measured data are fitted to LQ model.
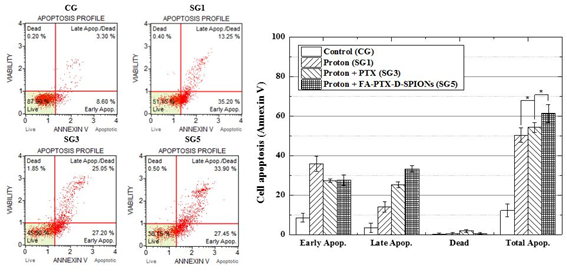
Figure 9. The results of Annexin V assays: left graphs show flow-cytometry analysis with Annexin V on proton beam irradiated C6 cells (SG1, SG3 and SG5) and without irradiation (CG). Right graph represents cell apoptosis of each group (Annexin V). The data present as the mean ± SD (n¼3). *p<0.05.
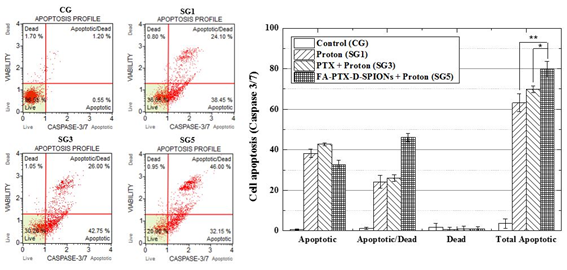
Figure 10. The results of capase 3/7 assays: left graphs show flow-cytometry analysis with caspase 3/7 on proton beam-irradiated C6 cells (SG1, SG3 and SG5) and without irradiation (CG). Right graph represents cell apoptosis of each group (caspase 3/7). The data present as the mean ± SD (n¼3). *p<0.05.
Abstract
PURPOSE:
This report presents novel nanoparticle-based drug delivery system (NPDDS) aiming to targeting chemo-proton therapy (TCPT) to improve the therapeutic efficacy on brain cancer treatments.MATERIALS AND METHODS:
A NPDDS, superparamagnetic iron oxide nanoparticles conjugated with folate and paclitaxel, was synthesized and applied to C6 brain cancer cell line that was prepared for targeting chemo-proton therapy. The characterization of NPDDS was analyzed by transmission electron microscope (TEM) and Fourier transform infrared (FTIR) spectroscopy. The uptake of NPDDS into the cytoplasm of C6 cells was observed by confocal laser scanning microscopy (CLSM). The therapeutic efficacy of proton beam was quantitatively evaluated by flow cytometry and clonogenic assay at various radiation dose.RESULTS:
NPDDS was synthesized in the uniform size distribution with a mean diameter of 5.44 ± 0.70 nm, and it showed no significant cytotoxicity at the concentration lower than 200 ng/mL. Radiosensitization enhancement factors of PTX, D-SPIONs and FA-PTX-D-SPIONs were 1.35, 1.16 and 1.52, respectively.CONCLUSIONS:
It was demonstrated that TCPT improved the therapeutic efficacy of the proton beam therapy when the synthesized novel NPDDS was administrated. The improvement in therapeutic efficacy was achieved by the synergetic effect of drug delivery increased by FA, radiosensitivity increased by PTX and absorption of proton energy increased by SPIONs.
Author informationKang SH1, Hong SP1, Kang BS1.
1
a Department of Radiological Science , Konyang University , Daejeon , South Korea.
- 키워드
- drug delivery system (DDS); folic acid (FA); paclitaxel (PTX); superparamagnetic iron oxide nanoparticles; targeting chemo-proton therapy
- 연구소개
- 방사선저항성이 높은 뇌암(glioblastoma multiforme)치료를 위해 입자빔에 의한 국부적 PISR(particle induced secondary radiation) 발생으로 방사선치료효과를 높임과 동시에 항암치료제를 이용한 화학치료가 가능하도록 금속나노입자-종양표적물질-항암제를 conjugation한 Targeting Chemo-Proton Therapy (TCPT) 약물전달시스템 개발 및 이를 이용한 뇌암치료효과 평가/분석에 관한 연구논문입니다. 개발된 TCPT DDS를 이용할 경우 약물의 암세포 집적도가 최대 2배 증가하고 암세포 생존률 1%에 대한 치료선량이 최대 25% 감소되어 뇌암 치료효과가 획기적으로 개선됨을 확인하였습니다. 최근 방사선 종양치료를 위해 많은 관심을 모으고 있는 입자빔 치료와 방사선저항성 암치료기술 및 약물전달시스템에 관련된 연구를 수행하는 연구자들에게 좋은 참고자료가 될 수 있을 것입니다.
- 덧글달기