글로벌 연구동향
방사선생물학
- 2019년 03월호
[J Clin Med.] Atrovastatin을 활용한 trastuzumab 부작용 저해연구Trastuzumab Induced Chemobrain, Atorvastatin Rescued Chemobrain with Enhanced Anticancer Effect and without Hair Loss-Side Effect.KIRAMS / 이선화, 이해준, 김진수*
- 출처
- J Clin Med.
- 등재일
- 2019 Feb 11
- 저널이슈번호
- 8(2). pii: E234. doi: 10.3390/jcm8020234.
- 내용
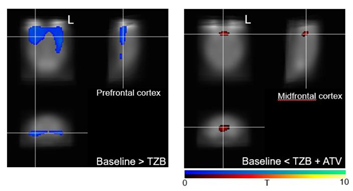
<그림> 항암표적치료제인 트라스투주맙 투여 시 뇌전두엽의 포도당대사가 감소하였고, 아토르바스타틴을 동시에 투여하였을 때 뇌 전두엽의 포도당 대사가 다시 회복되었다.
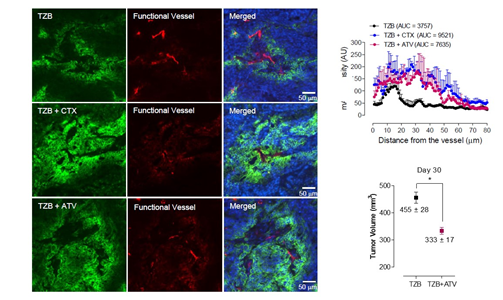
<그림> 항암표적치료제인 트라스투주맙 치료 시 아토르바스타틴을 동시에 투여하였을 때 종양에 섭취 되는 트라스투주맙 양이 증가하였고, 종양크기도 감소하였다.
Abstract
The authors identified that chemo-brain was induced after trastuzumab (TZB) therapy. In addition, atorvastatin (ATV) could rescue chemo-brain during trastuzumab (TZB) therapy. Enhanced therapeutic effect of TZB was confirmed after ATV therapy. We also investigated that there was no hair loss side effect due to ATV therapy. In an animal model, 150 μg TZB and five serial doses of 20 mg/kg ATV were administered. 18F-fluorodeoxyglucose Positron Emission Tomography (PET) and Magnetic Resonance Imaging (MRI) data were acquired. Statistical parametric mapping analysis and voxel-based morphometry analysis were performed to identify differences in glucose metabolism and gray matter concentration. The enhanced therapeutic efficacy of TZB after ATV treatment was assessed using a human epidermal growth factor receptor 2-positive gastric cancer model. We found a decrease in cerebral glucose metabolism and gray matter concentration in the frontal lobe following TZB therapy (p < 0.005). After subsequent ATV administration, glucose metabolism and regional gray matter concentration were rescued (p < 0.005). Cognitive impairment due to TZB and the rescue effect of ATV were confirmed using a passive avoidance test and quantitative real-time reverse transcription PCR. Furthermore, the penetration and accumulation of TZB in tumors increased by 100% after ATV co-administration, which resulted in an enhanced anti-cancer effect. Our study collectively demonstrates that ATV co-administration with TZB rescued the TZB-induced chemo-brain and enhances the therapeutic efficacy of TZB in tumors. We also showed that there was no hair loss during ATV therapy.
Author informationLee S1,2, Lee HJ3, Kang H4,5, Kim EH6, Lim YC7,8, Park H6, Lim SM9, Lee YJ10, Kim JM11, Kim JS12,13.
1
Division of RI application, Korea Institute of Radiological and Medical Sciences, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. remnant8724@naver.com.
2
Department of Bio-convergence Engineering, Korea University, Seoul 02856, Korea. remnant8724@naver.com.
3
Division of radiation biomedical research, Korea Institute of Radiological and Medical Sciences, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. hjlee@kirams.re.kr.
4
Division of RI application, Korea Institute of Radiological and Medical Sciences, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. hyunji.k0618@gmail.com.
5
Radiological and Medico-Oncological Sciences, University of Science and Technology, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. hyunji.k0618@gmail.com.
6
Department of Bio-convergence Engineering, Korea University, Seoul 02856, Korea. eh140149@kirams.re.kr.
7
Division of RI application, Korea Institute of Radiological and Medical Sciences, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. zerofe0701@gmail.com.
8
Research support team, ANDIVA Inc., Chuncheon 24324, Korea. zerofe0701@gmail.com.
9
Department of Nuclear Medicine, Korea Institute Radiological and Medical Sciences, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. smlim328@kcch.re.kr.
10
Division of RI application, Korea Institute of Radiological and Medical Sciences, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. yjlee@kirams.re.kr.
11
Department of Bio-convergence Engineering, Korea University, Seoul 02856, Korea. minbogun@korea.ac.kr.
12
Division of RI application, Korea Institute of Radiological and Medical Sciences, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. kjs@kirams.re.kr.
13
Radiological and Medico-Oncological Sciences, University of Science and Technology, 75 Nowon-ro, Nowon-gu, Seoul 01812, Korea. kjs@kirams.re.kr.
- 키워드
- anti-cancer effect; atorvastatin; chemo-brain; radiomics; trastuzumab therapy
- 연구소개
- 인지기능 저하는 항암치료 중 일시적으로 발생할 수 있는 부작용으로 원인을 밝히기 위한 다양한 연구가 진행되고 있다. 연구팀은 Her2 가 발현하는 암세포를 공격하는 표적치료제 트라스투주맙(trastuzumab)을 투여하고, 동시에 고지혈증 치료제인 아토르바스타틴(atorvastatin)을 투약한 후, 양전자방출단층촬영(PET/CT) 및 자기공명영상(MRI)을 통해 뇌 전두엽의 포도당 대사 및 뇌 부피를 비교 관찰하였다. 실험결과, 트라스투주맙을 단독으로 투여했을 때 전두엽 영역에서 포도당 대사와 부피가 감소하는 반면, 트라스투주맙과 아토르바스타틴을 동시에 투여한 경우 포도당 대사와 뇌 부피가 정상 수준으로 유지됨을 뇌 영상 통계기법으로 확인하였다. 항종양효과 측면에서도 아토르바스타틴을 트라스투주맙과 동시에 투여하였을 때트라스투주맙 단독 투여에 비하여 종양크기가 36% 더 감소하는 것을 확인하였다.
- 덧글달기







